 “If I waited till I felt like writing, I’d never write at all.” — Anne Tyler
“If I waited till I felt like writing, I’d never write at all.” — Anne Tyler
It’s been a couple of months since I last wrote a note or posted a picture. Thank you to the friends and followers who wrote asking if everything was okay: It is.
There has been a lot going on since April 22, milestones and changes, good times and difficult intervals. And the further I fell behind in sharing it all, the more difficult it became to start again.
Still, I made notes to myself, collected pictures and bookmarked articles, banking content for the future.
So, pen to paper and back to it.
I’ll likely backfill a bit, certainly look forward, and update on the many changes in life as time allows. Some of the constants are still there: I can be found in the same haunts in Poole and Maastricht, for now. My w.wezen and my medtech /entrepreneurial dreams remain my passions. I remain somewhat FOMO’d on weekends and occasionally tortuous under questioning.
But life is generally good, even if transitioning yet again.



 It sets out best practices for building a medical product, including design process, control of manufacture, human resources, and business organization. In each EU Member Country, the Competent Authority of experts sets the rules for each type of product in accord with these guiding principles. Then private companies, the Notified Body, assesses businesses and grant permission to market product by affixing a CE mark.
It sets out best practices for building a medical product, including design process, control of manufacture, human resources, and business organization. In each EU Member Country, the Competent Authority of experts sets the rules for each type of product in accord with these guiding principles. Then private companies, the Notified Body, assesses businesses and grant permission to market product by affixing a CE mark.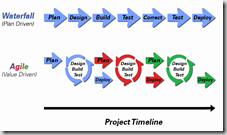


 a half a dozen people and six months to build it from nothing. The odds are heavily against passing on the first try in any case, much less with so little to apply to the task.
a half a dozen people and six months to build it from nothing. The odds are heavily against passing on the first try in any case, much less with so little to apply to the task. 












